Is Baso4 Ionic Or Covalent
Is a bond ionic or covalent?
Introduction:
An ionic bond is where ane cantlet takes one valence electron from the other. This volition give one atom a positive formal charge and the other a negative formal accuse. The two atoms stay together because of the electrostatic attraction of the plus and minus charges.
A covalent bond is where two atoms share ii electrons. They exercise this to try to fill their valence shells. Covalent bonds have well defined lengths (~one.0 to ii.five Å) and bail strengths (~100 kcal/mol).
How to:
Ane way to predict whether a bail is ionic or covalent is to look how far apart the two atoms forming the bonds are in the periodic table. If 1 atom is on the far left (Group ane or 2) and the other is on the far correct (Group 5, 6, or seven), then the atoms will take big differences in EN and will form an ionic bond. Most other pairs of atoms are close enough in EN to grade covalent bonds. For instance, any ii atoms in the main group elements (Groups 3, 4, v, 6, 7), will commonly form a covalent bond. Retrieve to treat hydrogen similar a Group 3 element as its EN is closest to boron.
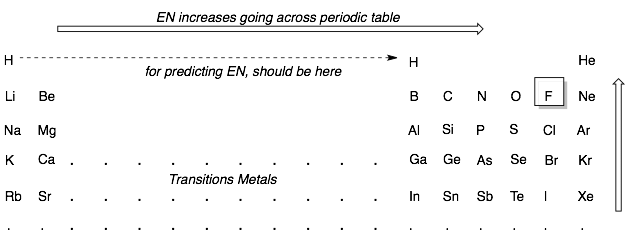
Examples:
Na and Cl –> ionic
K and O –> ionic
Ca and O –> ionic
C and F –> covalent
Si and Cl –> covalent
C and C –> covalent
C and H –> covalent
B and F –> covalent
Avant-garde:
Covalent and ionic are merely the 2 extremes of the different types of bonds. There is actually a continuum of bail types. Most bonds accept some covalent and some ionic character. For case, a C-C covalent bail has fiddling or no ionic character because the 2 atoms accept the same EN'southward. The Si-F is one of the most polarized covalent bonds and has a lot of ionic character. The N-Li bond is ionic only has meaning covalent graphic symbol.
Is Baso4 Ionic Or Covalent,
Source: https://shimizu-uofsc.net/orgo/knowledge-base/is-a-bond-ionic-or-covalent/
Posted by: mapplocies.blogspot.com


0 Response to "Is Baso4 Ionic Or Covalent"
Post a Comment